A Note from the President
TransPharm has given me some wonderful opportunities over the last 4 years. Locally, we volunteer for Junior Achievement, Big Brothers/Big Sisters, and donate to numerous charities that I wouldn’t have been able to in the past.
At the state level, we were recognized as one of the Top 50 Companies to Watch in Michigan in 2011. I was invited to a business round table discussion and met with Jennifer Granholm, Michigan’s outgoing Governor. I was also invited to Governor Rick Snyder’s residence in Lansing for the Moores River Drive Breakfast. Both events brought together many of the state’s business leaders and gave us a chance to meet with some of our state representatives to hear some of the new initiatives that are in the works for small business owners.
At the national level, TransPharm has traveled to both the East and West Coasts and many cities in between to attend the annual ICAAC conference. This year’s event is being held in San Francisco, California. Please stop by booth 1014 and say hello.
I feel extremely lucky to have traveled to London, England to attend the European Congress of Clinical Microbiology and Infectious Diseases. The conference boasted attendance from 106 different countries. It was my first trip to Europe and I can’t overstate how wonderful it was. Ironically, I met a family in our hotel that lives about 25 miles from me in Michigan. It just goes to show how small this world is these days. I’m already looking forward to attending the conference next year in Berlin, Germany.
Daniel Ross
President and CEO
TransPharm’s Development Incubator
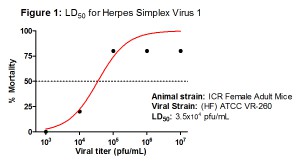
To update you on our current model development here at TransPharm, I’d like to announce that our first viral sepsis model, Herpes Simplex Virus 1, is just about completed! Once the finishing touches are added within the next few days we’ll be ready to roll this out to all our interested clients (Figure 1). Also while the viral models are emerging from our development incubator, we’ve also established a neonatal rat sepsis model using Staphylococcus epidermidis (Figure 2). This model mimics the clinical manifestation sometimes seen in low birth weight human infections.
In recent months, a new, advantageous partnership has emerged between TransPharm and HuMurine Technologies (www.humurine.com). Using their proprietary Hu-M™ CIEA NOG mouse® model as a background, HuMurine has developed a commercialized humanized mouse, named the Hu-M™ ID Model, to study infectious disease. With their novel humanized mouse model and TransPharm’s preclinical testing experience, this joint venture has the potential to greatly benefit institutions that desire to test compound efficacy in a preclinical, human immunological environment. We are looking to develop a hu-mouse model for all current TransPharm pathogens, as well as new ones such as Malaria and Dengue. Stay tuned for updates on this exiting, new partnership!
Dr. Santiago R. Lopez
Director of Research
Dan Hits the Airwaves!
Dan Ross, President & CEO of TransPharm, was recently interviewed by the Michigan Business Network. Click the link below to listen to the podcast.
http://www.michiganbusinessnetwork.com/radio/2012/03/09/Business_Beat_S2_Daniel_Ross
As always, please feel free to contact us:
Through our UPDATED website, www.transpharmsite.com
Our toll free INFECT line, 1-888-88-INFECT
Or dial me direct, 1-517-536-8210
Check us out on Facebook